What Is Hybridisation?
What Is Hybridisation?
In chemistry , hybridisation (or hybridization ) is the concept of mixing atomic orbitals into new hybrid orbitals (with different energies, shapes, etc., than the component atomic orbitals) suitable for the pairing of electrons to form chemical bonds in valence bond theory . Hybrid orbitals are very useful in the explanation of molecular geometry and atomic bonding properties.
Types of Hybridisation
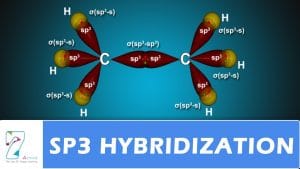
sp3
Four sp 3 orbitals.
Hybridisation describes the bonding atoms from an atom’s point of view. For a tetrahedrally coordinated carbon (e.g.,
methane CH 4 ), the carbon should have 4 orbitals with the correct symmetry to bond to the 4 hydrogen atoms.
Carbon’s ground state configuration is 1s 2 2s 2 2p 2 or more easily read:
C ↑↓ ↑↓ ↑ ↑
1s 2s 2p 2p 2p
The carbon atom can use its two singly occupied p-type orbitals, to form two covalent bonds with two hydrogen atoms, yielding the singlet methylene CH 2, the simplest carbene . The carbon atom can also bond to four hydrogen atoms by an excitation of an electron from the doubly occupied 2s orbital to the empty 2p orbital, producing four singly occupied orbitals.
C* ↑↓ ↑ ↑ ↑ ↑
1s 2s 2p 2p 2p
The energy released by formation of two additional bonds more than compensates for the excitation energy required, energetically favouring the formation of four C-H bonds.
Quantum mechanically, the lowest energy is obtained if the four bonds are equivalent, which requires that they are formed from equivalent orbitals on the carbon. A set of four equivalent orbitals can be obtained that are linear combinations of the valence-shell (core orbitals are almost never involved in bonding) s and p wave functions,[5] which are the four sp3 hybrids.
C* ↑↓ ↑ ↑ ↑ ↑
1s sp3 sp3 sp3 sp3
In CH 4 , four sp3 hybrid orbitals are overlapped by hydrogen 1s orbitals, yielding four σ (sigma) bonds (that is, four single covalent bonds) of equal length and strength.
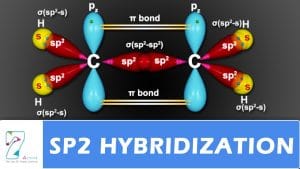
sp2
Three sp2 orbitals.
Ethene structure
Other carbon based compounds and other molecules may be explained in a similar way. For example, ethene (C 2 H 4 ) has a double bond between the carbons.
For this molecule, carbon sp 2 hybridises, because one π (pi) bond is required for the
double bond between the carbons and only three σ bonds are formed per carbon atom. In sp2 hybridisation the 2s orbital is mixed with only two of the three available 2p orbitals,
C* ↑↓ ↑ ↑ ↑ ↑
1s sp2 sp2 sp2 2p
forming a total of three sp 2 orbitals with one remaining p orbital. In ethylene (ethene ) the two carbon atoms form a σ bond by overlapping two sp 2 orbitals and each carbon atom forms two covalent bonds with hydrogen by s–sp 2 overlap all with 120° angles. The π bond between the carbon atoms perpendicular to the molecular plane is formed by 2p–2p overlap. The hydrogen–carbon bonds are all of equal strength and length, in agreement with experimental data.What Is Hybridisation?
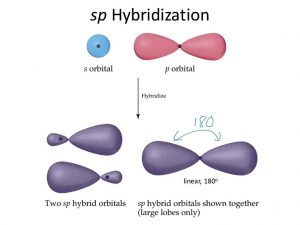
sp
Two sp orbitals
The chemical bonding in compounds such as
alkynes with triple bonds is explained by sp hybridisation. In this model, the 2s orbital is mixed with only one of the three p orbitals,
C* ↑↓ ↑ ↑ ↑ ↑
1s sp sp 2p 2p
resulting in two sp orbitals and two remaining p orbitals. The chemical bonding in acetylene (ethyne) (C 2 H 2 ) consists of sp–sp overlap between the two carbon atoms forming a σ bond and two additional π bonds formed by p–p overlap. Each carbon also bonds to hydrogen in a σ s–sp overlap at 180° angles.
Hybridisation and molecule shape
Hybridisation helps to explain molecule shape, since the angles between bonds are (approximately) equal to the angles between hybrid orbitals, as explained above for the tetrahedral geometry of methane. As another example, the three sp 2 hybrid orbitals are at angles of 120° to each other, so this hybridisation favours trigonal planar molecular geometry with bond angles of 120°. Other examples are given in the table below.For more info like this, visit: http://www.applygist.com
Related
Discover more from Applygist Tech News
Subscribe to get the latest posts sent to your email.